
To install it, unzip the archive and copy the octaveX directory in the application plugins folder, alongside all the plugins that come with the standard Texmacs installation. If you are interested to test it, you can download the zipped archive from here. I tried it on Windows, OSX and Linux, works on the systems I have access to. 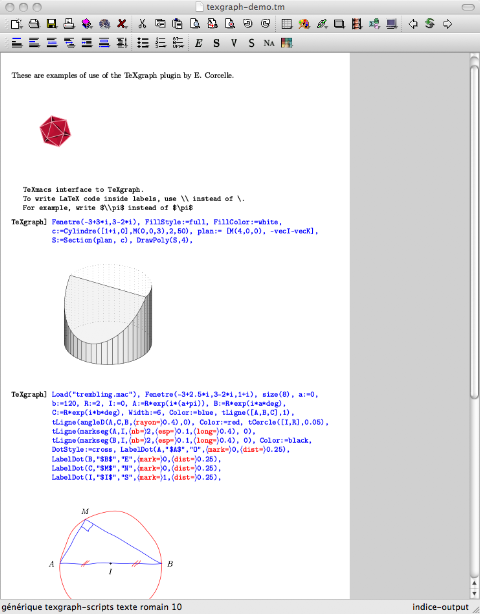
In addition to bringing practical solutions in the improvement of LV vector production, this work serves as a stepping stone for newcomers in the field as it highlights the critical variables needed to establish a cost-effective production process of LV vectors for research and early clinical studies on CAR-T cells. Quantification of CAR-T cells by flow cytometry demonstrated that transduction with both LV supernatants produced in serum-free medium (L-TEX and PJ-TEX) more than doubled the frequency of CAR+ cells compared to the L-DMEM condition (L-DMEM = 30.77%☐.04%, L-TEX = 76.01%☐.25%, PJ-TEX = 73.54%☑.05% CAR+cells). LV produced by Lipofectamine transfection and harvested in DMEM 10% FBS (L-DMEM group) were used as controls. To that end, we generated CAR-T cells by transducing human T-cells with LV particles harvested in TexMACs medium after transfection with Lipofectamine (L-TEX group) or PEI JetOptimus (PJ-TEX group). Finally, we evaluated if these tested variables in producing LV particles would affect transduction efficiency. Viral titer resulting of lipofectamine-based transfection in the same conditions was equivalent (1.29×10 7 ± 1.30×10 6 IU/mL). The yields were further increased when the DNA amount was reduced to 104 ng/cm 2 and the producer cell line was changed to LentiX-293T (1.32×10 7 ± 0.13×10 7 TU/mL). We found that among the PEI formulations, transfection with PEI JetOptimus resulted in the highest LV titers. In addition, viral titers were decreased as the amount of PEI was increased. The infectious titers obtained from calcium phosphate transfection method were the lowest among the tested methods. The resulting LV titers were compared to that obtained with lipofectamine-based transfection. Next, we compared the three most used DNA-delivery methods: calcium phosphate precipitation, cationic lipids (Lifofectamine™) and polycations (linear PEI 25 KDa, PEIPro and PEI JetOptimus). Among the media tested, harvesting in TexMACS and DMEM 10% FBS yielded the highest viral titers (4×10 6 transducing units per mL or TU/mL). 
For this purpose, HEK293/T17 cells were transfected with lipofectamine and viral titers were measured by flow cytometry. In addition, we included DMEM 10% FBS and FreeStyle media which are commonly used for viral production for comparison. Our hypothesis was that collecting LV particles in a medium optimized for T-cell growth, such as TexMACS and X-VIVO 10, would improve transduction efficiencies. Initially, we evaluated the LV titers harvesting in different media: TexMACS™, X-VIVO™10, FreeStyle™ and DMEM supplemented with 10% fetal bovine serum (FBS). Here, we demonstrated that an improvement in lentiviral production can be achieved by optimizing parameters such as serum-free media for lentivirus (LV) harvesting, PEI formulations, DNA amount, PEI:DNA ratios and producer cell lines. 
Optimizing the production of vectors for CAR-T cell transduction before beginning large-scale manufacturing reduces variability and maximizes efficiency. Production of CAR-T cells requires several carefully performed steps and consistent production of optimal viral vectors is a critical step to ensure efficient gene modification of lymphocytes. Chimeric antigen receptor (CAR) T cell therapy is a new approach for treatment of cancer.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
